Oral Presentation Australian Diabetes Society and the Australian Diabetes Educators Association Annual Scientific Meeting 2016
Earlier onset of exposure and greater early pharmacokinetic and pharmacodynamic effects for faster-acting insulin aspart vs. insulin aspart using continuous subcutaneous insulin infusion (#162)
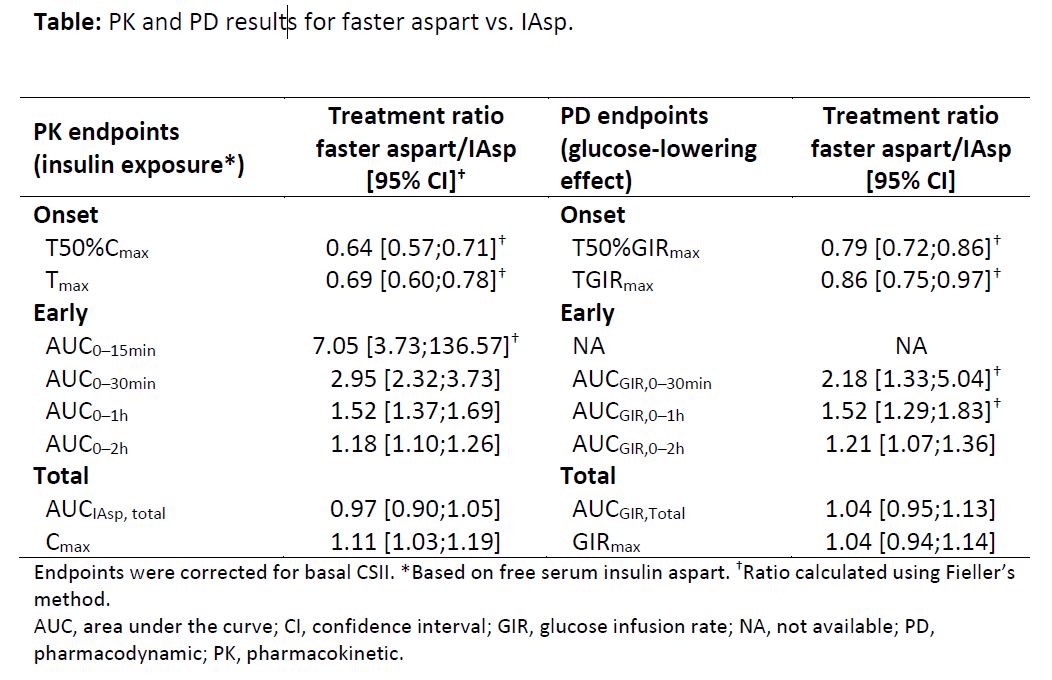
Background: faster-acting insulin aspart (faster aspart) is insulin aspart (IAsp) set in a new formulation with a faster initial absorption following subcutaneous injection. This trial evaluated the pharmacokinetic (PK) and pharmacodynamic (PD) properties of faster aspart during continuous subcutaneous insulin infusion (CSII) in a randomised crossover design.
Methods: patients (n=48) with type 1 diabetes (mean±SD age: 46.3±8.6 years; HbA1c: 7.4±0.6%) received faster aspart or IAsp as a CSII bolus dose (0.15 U/kg) on top of basal CSII (0.02 U/kg/h) under glucose-clamp conditions (blood glucose target 5.5 mmol/l [100 mg/dL]; total duration 27 h: 13 h run-in and 14 h post-bolus dosing).
Results: following a bolus dose, t50%Cmax and tmax occurred 36% (12 min) and 31% (26 min) earlier. Early insulin exposure (within the first 2 h) was greater for faster aspart than IAsp, with a near threefold greater exposure in the first 30 min (Table); total exposure was similar. Faster aspart had an earlier t50%GIRmax (21%; 11 min), tGIRmax (14%; 19 min) and a greater glucose-lowering effect during the first 2 h following bolus dosing vs. IAsp; total glucose-lowering effect was similar (Table). Both treatments were well tolerated.
Conclusion: faster aspart showed an enhanced early exposure and action compared with IAsp in CSII. Improvements in onset of exposure and action were more pronounced than those previously reported for subcutaneous injection of faster aspart.1
- Heise et al. Diabetes Obes Metab 2015;17:682–8